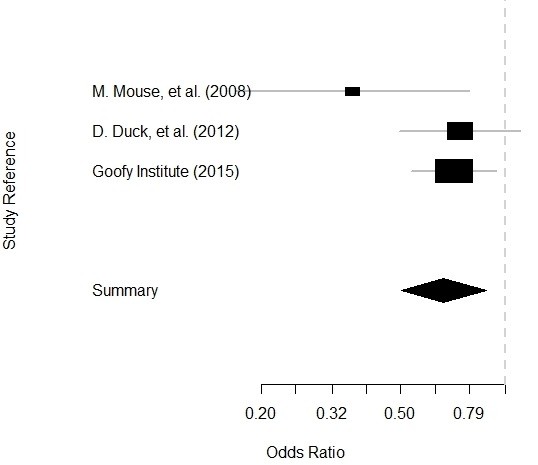
Getting data for meta-analysis together can be challenging, so I walk you through the simple steps I take, starting with the scientific literature, and ending with a gorgeous and evidence-based Forrest plot!
Tag Archives: research ethics
The United States (US) Center for Disease Control and Prevention (CDC) was in the spotlight during the COVID-19 pandemic. But as I describe in this blog post, it is only as good as its director.
I describe the three steps of my alternative model to the Plan-Do-Study-Act (PDSA) model for quality assurance/quality improvement (QA/QI) in healthcare.
Read my last post in a series on data-related misconduct at startup Theranos outlined in the book, “Bad Blood”, where I discuss their lack of administrative barrier between research and clinical data.
As a data science leader, what should you put in place so your organization doesn’t end up a data mess like startup Theranos? This blog posts provides guidance.
The Plan-Do-Study-Act model is promoted for quality assurance/quality improvement in healthcare. But does it have any peer-reviewed evidence base behind it? I examine that in this blog post.
This blog post talks about how lack of product description led to data-related misconduct at Theranos, because they could never nail down exactly what they were trying to do.
This is my first blog post in a series of five where I talk about data-related misconduct outlined in the book “Bad Blood”, and provide guidance on how to prevent it.